Germs are spread in a multitude of ways – from person-to-person, from food-to-person or from surfaces-to-person.1,2 When people visit public areas outside the home, such as foodservice facilities, they can come in contact with many surfaces, including tabletops, menus and highchairs. All of these surfaces are sources of moderate to high microbial contamination.3,4 Germs can persist in these areas for hours or even days. One study found that bacteria transferred to laminated menus persisted for six or more hours, posing a serious risk of cross-contamination.4
Another route for germ transmission is when people touch contaminated surfaces and fomites (pens, desks and other objects) and then touch their faces. Adults touch their face approximately 15 times an hour and about 360 times in a 24-hour period.5 Research has shown that people can touch surfaces an average of 301 times in hour and up to 3,600 times in 12 hours.3 You can imagine how that web can build. For harmful bacteria to spread, all it takes is for someone to touch a contaminated surface and then touch a door handle, elevator button, keypad, countertop, desk or other high-touch surface.
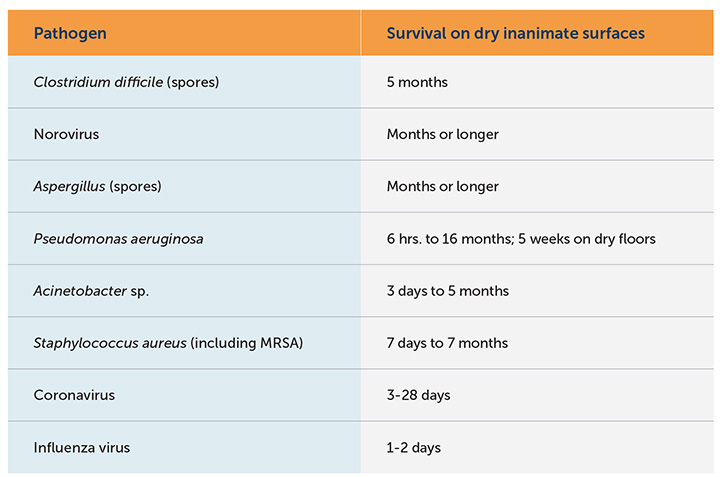
The chart above illustrates how bacteria and viruses can survive on dry surfaces for varying lengths of time, ranging from several hours to several months.4
To help break the cycle of germ transmission in away-from-home facilities where infection risk is heightened, it is critically important to identify the best ways to sanitize and disinfect surfaces.

Surface disinfection principles
To ensure proper surface disinfection, it’s essential to understand the distinctions between different methods and their effectiveness. It’s also essential to understand the differences between cleaning, sanitizing and disinfecting.
Antimicrobial pesticides are used on surfaces or non-living things and include wipes for kitchens, bathrooms and hospitals. These are regulated by the Environmental Protection Agency (EPA). Drugs and antiseptics, such as hand-sanitizing wipes, are used on living things. These are regulated by the U.S. Food and Drug Administration (FDA).
Antimicrobial agents are disinfectants and sanitizers that kill or slow the spread of microorganisms, including bacteria, viruses, protozoans and fungi, such as mold and mildew.6 The EPA defines a broad- spectrum disinfectant as one that can eliminate Staphylococcus aureus, Salmonella enterica or Pseudomonas aeruginosa within a maximum contact time of 10 minutes.7 Sanitizers are distinctly different with regard to EPA products. These products kill only bacteria, but have a decreased performance compared to an EPA disinfectant.
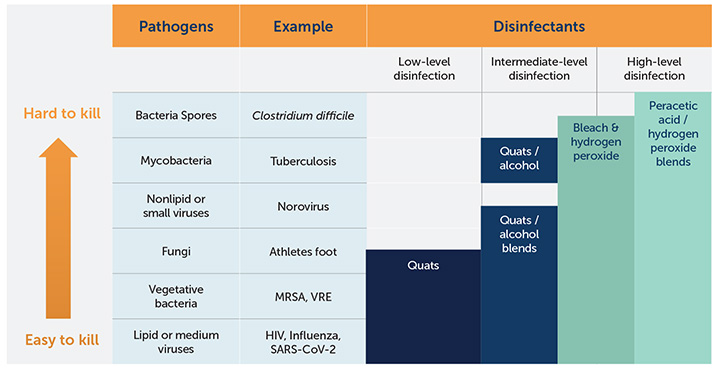
Factors that impact performance of all disinfectants are6,8,9:
- Concentration
- Active ingredients
- Time of exposure (contact time)
- Method of application (wipes, sprays, rags and bucket)
- Temperature
- pH
- Organic matter
The success of environmental surface disinfection is affected by cleaning procedures; use of appropriate tools; the volume and concentration of disinfectant applied to surfaces; disinfectant interaction with wipes, towels and mops; and, most of all, remembering to do it. By virtue of being pre-saturated, the disinfectant wipe helps eliminate a number of risks associated with human error such as improper dilution mix, over or under-spraying of surfaces and use of an incompatible substrate with the disinfectant chemical.
Residual hard surface antimicrobials
There are a variety of products available that sanitize and disinfect these surfaces. One major limitation of these products is that they fail to protect surfaces from subsequent contaminations throughout the day. Residual antimicrobials fill this gap.
The need for residual antimicrobials has been known for many years.10,11 Residual antimicrobials are sometimes referred to by other names, including persistent, durable and long-lasting antimicrobials. Some of the most common residual antimicrobials on durable surfaces contain metals such as zinc, copper and silver.12 Other durable surfaces use embedded biocides such as silane titanium oxides, quaternary ammonium compounds, as well as other polymers.13 There are also rechargeable durable coatings such as n-halamine.14 Fabrics are commonly treated with durable antimicrobials.15 The biocides added to durable finishes are affixed in a way to allow for a continual kill or static action for an extended period, some as long as the life of the treated product. A common characteristic for these types of treatments is that they are added to the article during manufacture and are not a user-applied treatment.
Many residual antimicrobial products are static in action vs. cidal (capable of killing bacteria). Static action will inhibit microbial growth but will not destroy it. Cidal action will destroy the organism. The most common uses for static antimicrobial solutions are odor control and maintaining product freshness.16
True long-term control of problem microbes requires a cidal (kill) action, or destruction of the microbes. For a residual antimicrobial to do this it must retain the ability to interact with the cell and be cidal. In addition, the antimicrobial must not only be a strong kill agent, but it must also be applied evenly – covering the entire surface – and be able to be retained and released on the surface after treatment. Durable treatments with metals and other biocides accomplish this via a slow release of biocidal agent into the environment that will kill the microbes. This is also true with other forms of residual antimicrobials including those that are applied to a surface by the user rather than during manufacture of the article.

With a user-applied residual product, the formulation is designed to produce a film on the surface that retains the antimicrobial, releasing it on demand. These products should also produce a film that is not visible to the user and has acceptable touch characteristics. Only a limited number of residual antimicrobial products have received EPA approval and nearly all are applied by spraying a solution onto a hard surface.
Spraying may not deliver an optimal level of a solution if the spray nozzle is faulty or if the end user applies an inadequate amount of liquid to the surface. In addition, end users may not wait for the recommended dwell time to be completed before wiping, which will reduce the level of effectiveness.17,18
User-applied residual antimicrobials have been around more than 60 years. The first reported use was in 1959 with the application of orthophenylphenol to hospital surfaces.19 In the early 2000s, a handful of user-applied hard surface disinfectant/sanitizer products with residual claims were introduced to the market. In comparison to the orthophenylphenol product from the 1950s, the most commonly listed actives for these products are cationic biocides20 or metals such as silver or copper.12 What all these products have in common is that they meet the EPA standards for residual self-sanitizers (RSS).21
The importance of meeting the performance recommendations outlined in the RSS method is that this method tests the residual product with conditions that try to remove it from the treated surface. For a product to be truly residual, it must be able to kill repeatedly for at least 24 hours while being resistant to attempted removal by wet and dry abrasions.21
In the United States, the EPA regulates sanitizers and disinfectants differently. Sanitizer products sold in the U.S. are specific to bacteria and have been proven to kill 3 log of bacteria on a surface in five minutes or less.7 Disinfectant products are required to show equal to or greater than 6 log killing of a bacteria, viruses or fungi from a surface in 10 minutes or less.7 The EPA residual self-sanitizing (RSS) protocol utilizes wear cycles (wet and dry abrasions) and microbial loading to demonstrate a product’s ability to remain on a surface and continue to kill bacteria to 99.9% for 24 hours. This protocol demonstrates the product’s durability and efficacy while simulating real-world wear and soiling. To assure broad-spectrum activity, the EPA currently requires the RSS test to be conducted with a representative Gram positive (Staphylococcus aureus) and Gram negative (Enterobacter aerogenes or Klebsiella pneumoniae) strain. The EPA recently defined the endpoints required to residually disinfect bacteria on a surface. The abrasion sequence is the same as for the RSS, but the endpoints require 5 log kill in at least 10 minutes rather than the 3 log required for an RSS endpoint.21
The primary advantage of residual sanitizers or disinfectants is that the surface can kill for an extended period of time, allowing for continuous protection from fomite transfer. Several studies have been done to demonstrate the benefits in actual use scenarios.22,23
There are a variety of products available that sanitize and disinfect these surfaces. One major limitation of these products is that they fail to protect surfaces from subsequent contaminations throughout the day. Residual antimicrobials fill this gap.
Surface antimicrobial wipes
Antimicrobials are commonly applied to a surface by two divergent methods – spraying or wiping. Users who apply spray products tend to combine wiping after spraying, adding some complexity to the process. Disinfection wipe products come in two forms – either a solution is added to a wipe, or a wipe is pre-saturated with an antimicrobial solution. Regardless of the product form or method of application, for a residual product to be most effective, a uniform coating should be applied to the entire surface. Therefore, it is important to understand how the disinfectant is applied. The remainder of this section focuses on the role a wipe plays in disinfecting surfaces.9,17,24
When it comes to wiping to deliver the antimicrobial, there are several options to choose from:
- Paper towels, which contain wood pulp
- Non-woven wipes
- Polymer-based towels
- Meltblown
- Spunbond
- Microfiber
- Mixed fiber wipes, which contain wood pulp and polymer
- Hydroknit
- Coform
- Polymer-based towels
- Woven towels, which contain cotton
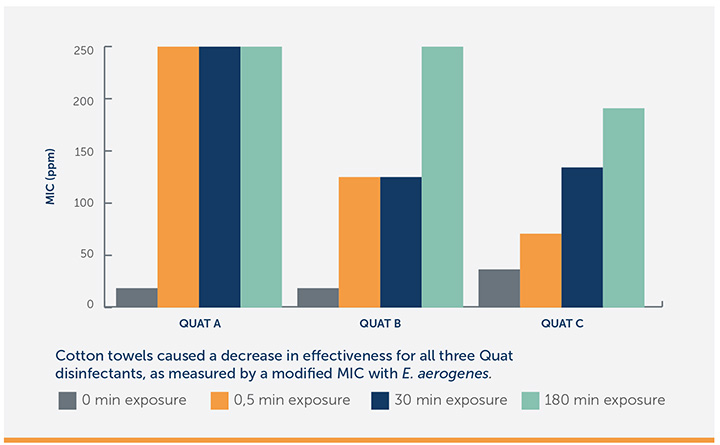
If you use a disinfectant system that adds the disinfectant to the wipe, it’s essential to make sure you use the right wiping material – one that is compatible with the disinfectants. Research has shown that the wiping material you use can dramatically affect the amount of disinfecting agent that reaches the surface being cleaned. A prime example: Quaternary Ammonium Compounds (a.k.a., Quats). Quats are attracted to and absorbed into fabrics, such as cotton towels.
A 2013 study in the American Journal of Infection Control found that cotton towels may reduce the effectiveness or even inactivate the ability of Quats to disinfect surfaces.25 The study found that laundered cotton towels soak up and hold disinfectant so that it doesn’t reach the surface at the recommended concentration level. As a result, cotton towels were found to reduce the disinfection strength of Quat-based disinfectants by up to 85%.
The use of pre-saturated wipes ensures that the wipe material is compatible with the “killing active” used in the disinfectant product. The most common antimicrobial actives used in residual products are Quats, which means that the wipes used to deliver this active must not contain cellulose or other negatively charged materials.25 In addition, pre-saturated wipes ensure the necessary volume of disinfectant is added to a surface to permit effective kill.
Wiping is also the best way to make sure the entire surface is treated. Wipes help reduce the risk of incomplete coverage as compared to sprays.
Knowledge is power
A study published in the American Journal of Infection Control demonstrates how both a hand and surface hygiene intervention significantly impacted virus transmission in a long-term care facility.26 The study found that education combined with the right solutions in the right locations greatly reduced the spread of viruses. As a result of the hygiene intervention, the number of viruses on surfaces was reduced by 99.9% and the presence of viruses on hands was reduced by 99%. The probability of the risk of infection from rhinovirus, influenza and norovirus was also significantly reduced due to the use of these interventions.
What this and the other studies cited in this paper demonstrate is that building a comprehensive hygiene and disinfection program that includes residual antimicrobials and, ideally, residual cidal wipes can make a significant difference in helping reduce the spread of pathogens.
When selecting solutions for your facility, it’s important to distinguish between static solutions, which inhibit the growth of microorganisms, and cidal solutions, which destroy microorganisms. For the best possible results, look for solutions that offer long-lasting residual protection and continue to protect surfaces even after multiple touches. In addition, consider the method for applying residual solutions. It is preferable not to use a cotton towel or a wipe that contains cellulose or other negatively charged fibers.25 A pre-saturated wipe with the appropriate base sheet technology will help ensure that the necessary volume of disinfectant is added to the surface to enable an effective kill. Wiping is also the best way to ensure that the entire surface is treated because it allows for complete coverage of complex surfaces and can reach areas that sprays may miss.
References
1. Kwok, Y.L.A., Gralton, J. and McLaws, M.L., 2015. Face touching: a frequent habit that has implications for hand hygiene. American Journal of Infection Control, 43(2), pp.112-114.
2. Lewis, R.C., Rauschenberger, R. and Kalmes, R., 2021. Hand-to-mouth and other hand-to-face touching behavior in a quasi-naturalistic study under controlled conditions. Journal of Toxicology and Environmental Health, Part A, 84(2), pp.49-55.
3. Zhang, N., Li, Y. and Huang, H., 2018. Surface touch and its network growth in a graduate student office. Indoor Air, 28 (6), pp.963-972.
4. Stephens, B., Azimi, P., Thoemmes, M.S., Heidarinejad, M., Allen, J.G. and Gilbert, J.A., 2019. Microbial exchange via fomites and implications for human health. Current Pollution Reports, 5(4), pp.198-213
5. Nicas, M. and Best, D., 2008. A study quantifying the hand-to-face contact rate and its potential application to predicting respiratory tract infection. Journal of occupational and environmental hygiene, 5(6), pp.347-352.
6. Block, S.S. ed., 2001. Disinfection, sterilization, and preservation. Lippincott Williams & Wilkins.
7. EPA. 2018. Series 810 – Product Performance Test Guidelines. https://www.epa.gov/test-guidelines-pesticides-and-toxic-substances/series-810-product- performance-test-guidelines. Accessed June 17, 2021.
8. Dvorak, G., 2005. Disinfection 101. Center for food security and public health, Iowa State University, Ames, IA.
9. Song, X., Vossebein, L. and Zille, A., 2019. Efficacy of disinfectant-impregnated wipes used for surface disinfection in hospitals: a review. Antimicrobial Resistance & Infection Control, 8(1), pp.1-14
10. Lester, W. and Dunklin, E.W., 1955. Residual Surface Disinfection: I. the Prolonged Germicidal Action of Dried Surfaces Treated with Orthophenylphenol. The Journal of Infectious Diseases, 96(1), pp.40-53.
11. Klarmann, E.G., Wright, E.S. and Shternov, V.A., 1953. Prolongation of the antibacterial potential of disinfected surfaces. Applied Microbiology, 1(1), p.19.
12. Brady, M.J., Lisay, C.M., Yurkovetskiy, A.V. and Sawan, S.P., 2003. Persistent silver disinfectant for the environmental control of pathogenic bacteria. American Journal of Infection Control, 31(4), pp.208-214.8 BMC microbiology, 14 (1), pp.1-14.
13. Huang, K.S., Yang, C.H., Huang, S.L., Chen, C.Y., Lu, Y.Y. and Lin, Y.S., 2016. Recent advances in antimicrobial polymers: a mini-review. International Journal of Molecular Sciences, 17(9), p.1578.
14. Liang, J., Wu, R., Wang, J.W., Barnes, K., Worley, S.D., Cho, U., Lee, J., Broughton, R.M. and Huang, T.S., 2007. N-halamine biocidal coatings. Journal of Industrial Microbiology and Biotechnology, 34 (2), pp.157-163.
15. Annis, P.A. ed., 2012. Understanding and Improving the Durability of Textiles. Elsevier.
16. McQueen, R.H. and Vaezafshar, S., 2020. Odor in textiles: A review of evaluation methods, fabric characteristics, and odor control technologies. Textile Research Journal, 90(9-10), pp.1157-1173.
17. Wiemken, T.L., Curran, D.R., Pacholski, E.B., Kelley, R.R., Abdelfattah, R.R., Carrico, R.M. and Ramirez, J.A., 2014. The value of ready-to-use disinfectant wipes: compliance, employee time, and costs. American Journal of Infection Control, 42(3), pp.329-330.
18. Rutala, W.A., Gergen, M.F. and Weber, D.J., 2012. Efficacy of different cleaning and disinfection methods against Clostridium difficile spores: importance of physical removal.
19. Dunklin, E.W. and Lester Jr, W., 1959. Residual Surface Disinfection: II. The Effect of Orthophenylphenol Treatment of the Floor on Bacterial Contamination in a Recovery Room. The Journal of Infectious Diseases, pp.41-55.
20. Zhou, C. and Wang, Y., 2020. Structure–activity relationship of cationic surfactants as antimicrobial agents. Current Opinion in Colloid & Interface Science, 45, pp.28-43.
21. EPA. 2015. Protocol for residual self-sanitizing activity of dried chemical residuals on hard, non-porous surfaces. Environmental Protection Agency website. https://www.epa.gov/sites/production/files/2015-09/documents/cloroxpcol_final.pdf. Accessed June 17, 2021.
22. Schmidt, M.G., Fairey, S.E. and Attaway, H.H., 2019. In situ evaluation of a persistent disinfectant provides continuous decontamination within the clinical environment. American Journal of Infection Control, 47(6), pp.732-734.
23. Rutala, W.A., Gergen, M.F., Sickbert-Bennett, E.E., Anderson, D.J., Weber, D.J. and CDC Prevention Epicenters Program, 2019. Antimicrobial activity of a continuously active disinfectant against healthcare pathogens. Infection Control & Hospital Epidemiology, 40(11), pp.1284-1286.17 Journal of Hospital Infection, 74 (1), pp.80-82.
24. Sattar, S.A. and Maillard, J.Y., 2013. The crucial role of wiping in decontamination of high-touch environmental surfaces: review of current status and directions for the future. American Journal of Infection Control, 41(5), pp. S97-S104.
25. Engelbrecht, K., Ambrose, D., Sifuentes, L., Gerba, C., Weart, I. and Koenig, D., 2013. Decreased activity of commercially available disinfectants containing quaternary ammonium compounds when exposed to cotton towels. American Journal of Infection Control, 41(10), pp.908-911.
26. Sassi, H.P., Sifuentes, L.Y., Koenig, D.W., Nichols, E., Clark-Greuel, J., Wong, L.F., McGrath, K., Gerba, C.P. and Reynolds, K.A., 2015. Control of the spread of viruses in a long-term care facility using hygiene protocols. American Journal of Infection Control, 43(7), pp.702-70.


